CrystalGenomics said it hoped to carry out a phase-1 clinical trial on new leukemia drug candidate CG-26806 (CG-806) in the first quarter of next year. The company is soon to submit the Investigational New Drug (IND) Application to the U.S. Food and Drug Administration.
CG-806 is a targeted therapy that inhibits bruton tyrosine kinase (BTK), FMS-like tyrosine kinase 3 (FLT3), and aurora kinase (AURK).
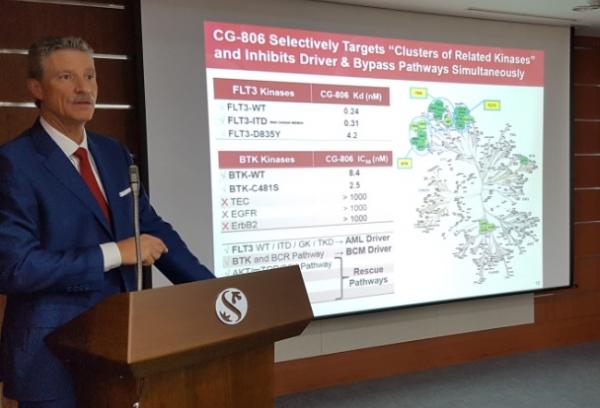
CrystalGenomics licensed out CG-806’s global sales rights, except for Korea and China, to Aptose Biosciences of the U.S. for 360 billion won ($319.1 million) in June 2016. In June, it additionally gave the exclusive rights for 134 billion won to develop and commercialize the drug within China.
Aptose Biosciences CEO William Rice, who will lead the study on CG-806, production, and commercialization (except for Korea), shared the progress of the drug candidate at a conference with analysts at the Shinhan Financial Investment building in Yeouido, Seoul, on Monday.
The FDA has suggested that since AML is an acute condition, Aptose should start the single ascending dose, pharmacokinetic (PK) study, of CG-806 in healthy volunteers in order to identify the therapeutic dose for AML patients, and take that dose directly into AML patients.
This would allow the sparing of very sick AML patients who are given non-therapeutic doses and potentially allow the company to seek clinical proof-of-concept at the first doses of the planned AML study.
As CG-806 showed a relatively high safety in preclinical tests, the company decided to conduct the upcoming phase-1 study on healthy people, Aptose said.
Aptose sees the FDA’s proposal as a positive signal for the development of CG-806. Deriving the right dose from healthy people can save time than doing so with cancer patients who have to start treatment with a low dose.
It is also positive news that if Aptose expands the indication of the drug for non-cell cancer and chronic lymphocytic leukemia, the company’s appropriate dose will be recognized, the company said.
“It is still a plan, but we are expecting to start the clinical phase in the first quarter of 2019,” Rice said. “We may have better clinical results with healthy people and save time. To date, both Aptose and CrystalGenomics have been satisfied with preclinical results.”
In December, CG-806 obtained the FDA’s orphan drug designation (ODD) in AML indication, which will reduce the time for trials.
Aptose said it would seek to win ODD for other diseases as well.
According to Rice, the U.S. recognizes a rare disease if the disease affects fewer than 200,000 people nationwide.
“The ODD requires evidence that a new treatment is superior to conventional drugs, and CG-806 proved this,” he said. “Although the experimental drug has ODD status for AML only, we will challenge to obtain ODD for non-cell cancers and chronic lymphocytic leukemia.”
The reason for the delay in the IND filing is "due to the additional studies being performed to enable us to dose healthy volunteers as a result of the favorable toxicity safety profile observed thus far," Rice said.
“CG-806 is an effective medicine but a compound that is very difficult to produce. We have resolved all the production issues, meeting the requirements for kilogram-based production,” he added.

