Chong Kun Dang said Monday that it has presented the clinical trial results for Cendom, an erectile dysfunction treatment, at the European Society for Sexual Medicine.
The trial presented at the conference relates to a nine-month study, conducted by Professor Yang Dae-yeol of the department of urological department at Hallym University Medical Center, on 102 patients, aged 20 to 70 years, who had a prostate problem with an International Prostate Symptom Score (IPSS) of eight or more and had a major postvoid dribble problem.
The researchers divided the patients into two groups, each receiving 12-week doses of Cendom or placebo, and then assessed the improvement of the postvoid dribble symptoms after urination.
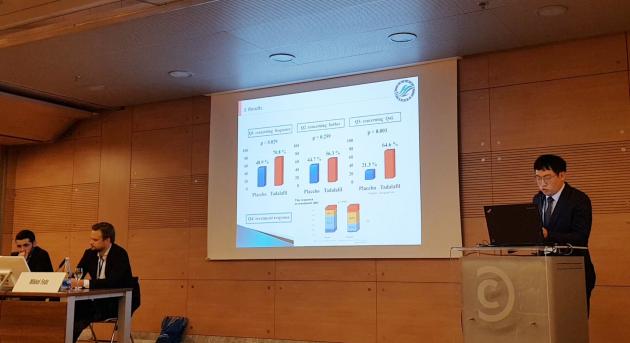
As a result, the research showed that the postvoid dribble levels in the Cendom group significantly improved compared to placebo groups.
Of the patients taking Cendom, 68.8 percent showed a significant improvement of more than 2 points in the international prostate symptom score compared to the 31.9 percent in the placebo group.
In the questionnaire after taking the drug, 68.7 percent of the patients taking Cendom responded that their symptoms improved.
The Cendom group also improved IPSS, and the International Index of Erectile Function compared to the placebo group, with no significant adverse events.
“Postvoid dribble is an important disease that interferes with the patient's daily life, but there are not many studies for treatment,” a company official said. “The results of this clinical study confirmed that the drug is effective in improving the symptomatic symptoms after going to the bathroom, and the company expects that the inconvenience to patients will sharply drop.”

