
As preannounced by Biogen in October, its Alzheimer's drug candidate aducanumab has achieved significant efficacy at high doses.
However, only one of the two phase 3 trials achieved p-values, and the high rate of discontinuation adverse reactions or amyloid-related imaging abnormalities compared to placebo has raised questions if the results can lead to an approval by the U.S. Food and Drug Administration.
Biogen presented the top-line results of its two phase 3 clinical studies -- EMERGE and ENGAGE -- for aducanumab at the Clinical Trials of Alzheimer's Disease (CTAD) conference in San Diego, Calif., last Thursday.
According to the EMERGE study, high doses of aducanumab in patients with early Alzheimer's disease showed a significant improvement in the Clinical Dementia Rating Scale Sum of the Boxes (CDR-SB). In March, the company announced that it would cease development into aducanumab due to a failure in the futility analysis in the two phase 3 clinical studies.
However, the company drew attention after revealing that the final analysis, including patients added after the data cutoff point, showed valid results in some high-dose groups, and that it plans to push ahead with the FDA approval process based on the results, unveiled during the CTAD conference.
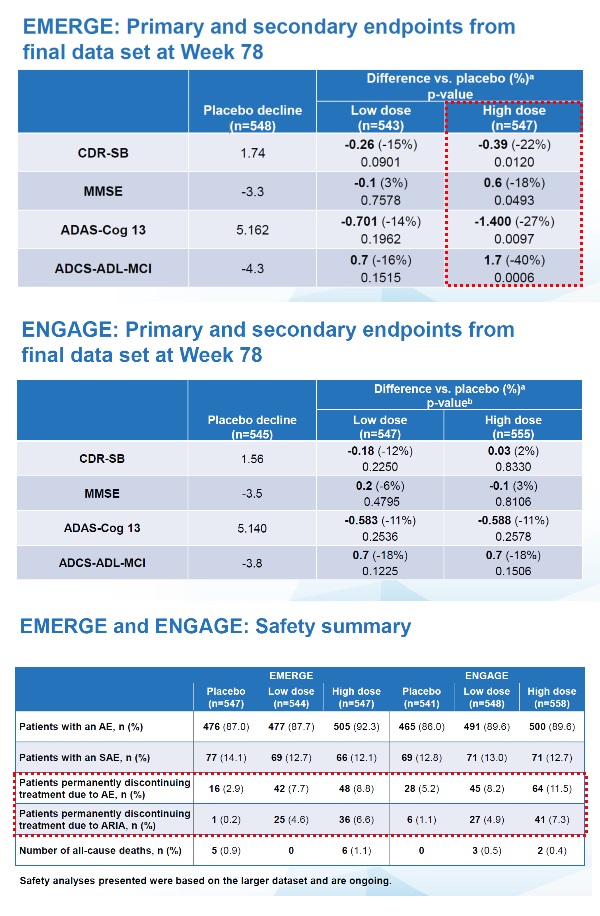
The EMERGE and ENGAGE studies evaluated 18 months of aducanumab treatment versus placebo in 3,285 early Alzheimer's patients enrolled in 348 locations in 20 countries. The primary endpoint was CDR-SB, and the secondary endpoint was the Mini-Mental State Examination (MMSE), Alzheimer's Disease Assessment Scale-Cognitive Subscale 13 (ADAS-Cog 13), and the Alzheimer's Disease Cooperative Study-Activities of Daily Living Inventory (ADCS-ADL-MCI) score.
Analysis of the final data at Week 78 showed that aducanumab achieved a primary endpoint in the high dose group within the EMERGE study. The average change in CDR-SB in the placebo group increased by 1.74 points compared to baseline. In contrast, the aducanumab high-dose group increased only 1.35 points, significantly lowering progression compared to placebo. Secondary endpoints also showed significant improvement in the EMERGE study.
However, the results of the ENGAGE study were different, as it failed to demonstrate efficacy for both primary and secondary endpoints.
Also, although the safety data has not yet been finalized, the expanded data analysis showed that the rate of discontinuation adverse reactions and amyloid-related visual abnormalities is remarkably high compared to placebo.
According to the industry officials, Biogen was reported to have issued a license application in October after discussing with the FDA from the moment it received valid results at high doses for aducanumab.
As there are no candidates that have been able to produce effective results for the treatment of Alzheimer's disease using anti-amyloid-mechanism to date, the industry is closely watching whether the U.S. regulator will hold Biogen’s hand only with the latest results.

